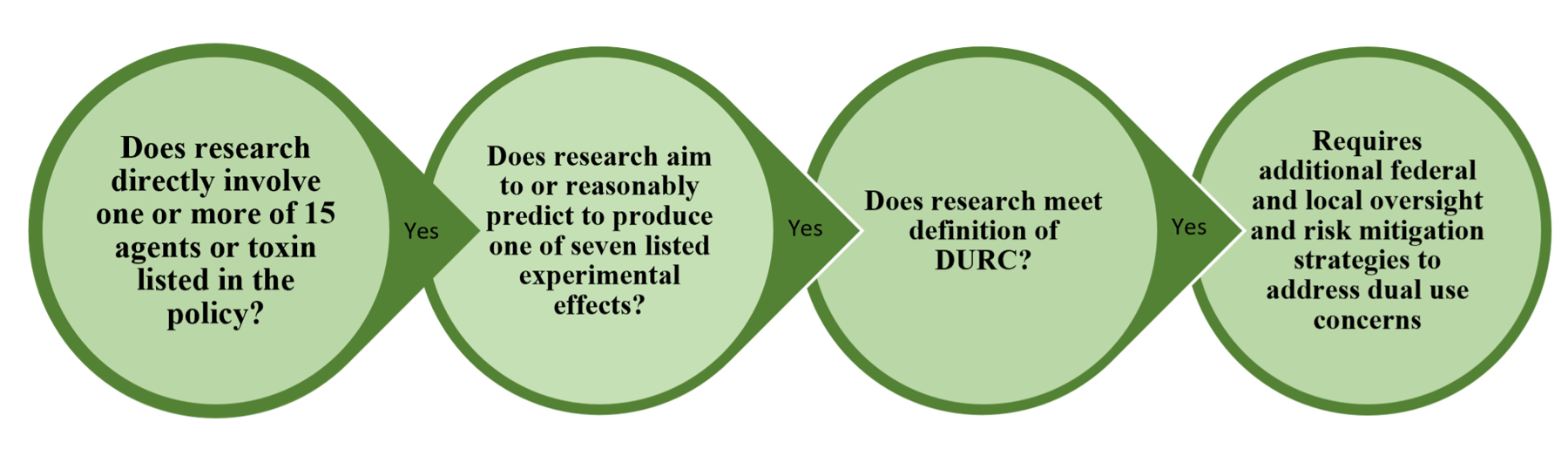
Can the research be reasonably anticipated to produce one or more of the seven experimental effects/categories listed below?
- Will an intermediate or final product of your research make a vaccine less effective or ineffective?
- Will the final or intermediate product of your research confer resistance to antibiotics or antivirals in ways that are inherently different than those published previously?
- Will your work enhance the virulence of a pathogen or render a non-pathogen virulent?
- Will the results of your work increase the transmissibility of any pathogen?
- Will your research result in alteration of the host range of a pathogen?
- Will your research result in a product or intermediate that that may prevent or interfere with diagnosis of infection or disease?
- Does your research enable “weaponization” of an agent or toxin?
- Even though your research did not involve any of the aforementioned seven criteria, and recognizing that your work product or results of your research could conceivably be misused, is there the potential for your results/product to be readily utilized to cause public harm?
- If the answer is "No", no further action is required, but the PI should conduct an ongoing assessment that this continues to be the case and must file an annual report of that assessment.
- If one or more of the seven experimental effects/categories listed above can potentially occur, the Institutional Biosafety Committee (IBC) and DURC working with the Pl assesses if the criteria defining DURC would potentially be met. Again, if the answer is "No", no further action is required, but the PI should conduct an ongoing assessment that this continues to be the case and must file an annual report of that assessment.
- If the criteria defining DURC would potentially be met, the DURC working with the PI must develop and implement a risk management plan based on the risk assessment. The conduct and or communication of the research findings must adhere to the risk management plan with ongoing oversight by the IBC and DURC with respect to DURC and in consultation with other UNT Committees as appropriate.