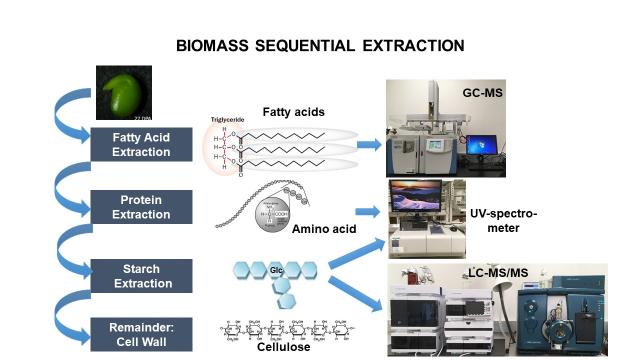
Macromolecules
Macromolecules (fatty acids, proteins, starch, and cell wall) are the end products of the metabolism of a living organism. In order to measure the quantity of biomass present in a specific micro-organism or tissue, a sequential biomass extraction is performed (see drawing). Detailed steps concerning the extraction and quantification of the macromolecules are reported in this pdf document.
Total Fatty Acids and Fatty Acids Composition (FAMEs)
Fatty acids are extracted and, then derivatized into Fatty Acid Methyl Esters (FAMEs) using methanol and sulfuric acid. The separation and detection of FAMEs is carried out using a 250 Omegawax (30 m x 0.25 mm x 0.25 micron) column from Supelco and the Thermo Trace 1310 Gas Chromatography Coupled to an ISQ Single Quadrupole Mass Spectrometer (Tsogtbaatar et al., 2015). The run time varies from 7 to 15 minutes.
Total Protein content
After fatty acid removal, proteins are extracted with a buffer, and then quantified using a colorimetric protein quantification kit from Bio-Rad. The reading is performed at 750 nm with a spectrophotometer.
Starch
After fatty acid and protein removal, starch is deconstructed using autoclaving and enzymatic steps. Then, a colorimetric starch quantification kit from Megazyme is utilized. The reading is performed at 510 nm with a spectrophotometer.
Proteinogenic Amino Acids
The composition of proteins can be assessed by hydrolysis with 6 N HCl at 125ºC for 24 hours. The resulting proteinogenic amino acids are analyzed using the LC-MS/MS method mentioned in the section "Amino Acids and Derivatives". The different steps associated with protein hydrolysis are reported in this pdf document.
Cell Wall
The analysis of cell wall, cellulose and hemicelluloses, is performed following a protocol published by Foster et al. (2010). After the removal of soluble metabolites, fatty acids, and starch; hemicelluloses are hydrolyzed into neutral sugars that are reduced and acetylated prior GC-MS analysis. A SP2380 (30 m x 0.25 mm x 0.20 micron) column from Supelco and a Thermo Trace 1310 Gas Chromatography Coupled to an ISQ Single Quadrupole Mass Spectrometer are utilized to resolve and quantify alditol acetates. The separation time is 25 minutes.
Crystalline cellulose is hydrolyzed using the Saeman hydrolysis. Then, the glucosyl residues from cellulose are quantified by the colorimetric anthrone assay. Absorption reading is made at 625 nm with a spectrophotometer.